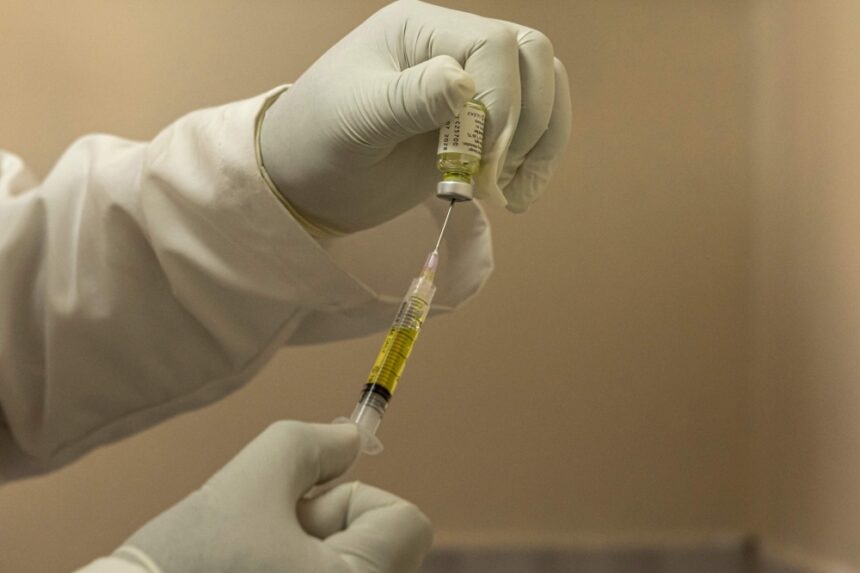
Nigeria’s health authorities have cautioned that the newly introduced long-acting HIV prevention injection, Lenacapavir (LenPrEP), should not be used by pregnant women. The Director-General of the National Agency for the Control of AIDS, Dr. Temitope Ilori, explained this during a media briefing organised by the National HIV, Viral Hepatitis and STDs Control Programme in Abuja.
Although international studies indicate that the drug is generally safe, experts say there is not enough evidence about how it may affect pregnancy, which is why its use in pregnant women is currently restricted. Officials also emphasised that Lenacapavir is designed to prevent HIV infection, not to treat people who already have the virus.
Health authorities described the injection as a major innovation in HIV prevention because it only needs to be administered twice a year, making it easier for people who struggle to take daily pills. However, they stressed that it does not protect against other sexually transmitted infections or diseases such as hepatitis, meaning individuals must still practise comprehensive sexual health precautions.
The National Coordinator of the AIDS and STDs Control Programme, Adebobola Bashorun, noted that the government plans to expand access to the drug across more states and vulnerable populations, with additional funding being explored to support wider distribution.
Officials further clarified that Lenacapavir will complement existing prevention methods rather than replace them. Options such as oral pre-exposure prophylaxis (PrEP) will remain available, and national guidelines will determine how the new injection fits into current programmes. Reported side effects so far have been mild, mainly limited to temporary pain at the injection site, with ongoing monitoring to ensure safety as rollout continues.
The Minister of State for Health, Dr. Iziaq Salako, stated that Nigeria is introducing the drug with support from the Global Fund and hopes to eventually engage manufacturers directly, including exploring possibilities for local production. Discussions around technology transfer and domestic manufacturing are seen as key to long-term sustainability and reducing dependence on foreign donors.
Lenacapavir, developed by Gilead Sciences, has drawn global attention as a potential breakthrough because of its long-acting protection against HIV. Nigeria carries one of the largest HIV burdens in the world, with about 1.9 million people living with the virus. While treatment access has improved significantly over the years, prevention gaps still exist, particularly among young people and high-risk groups.
Public health experts believe the new injection could significantly strengthen Nigeria’s HIV prevention strategy if it becomes widely accessible. However, concerns remain about affordability, long-term funding, and whether the healthcare system can support large-scale distribution without neglecting other critical health priorities such as malaria control, maternal care, and routine immunisation. For now, authorities view Lenacapavir as an important addition to existing prevention tools, but not a single solution to the country’s HIV challenge.
Lenacapavir itself is a long-acting antiviral drug designed to stop HIV from establishing infection in the body. It works by interfering with the virus’s ability to replicate and spread, offering extended protection from a single injection that lasts for months.
Unlike traditional daily prevention pills, this injection provides sustained drug levels in the bloodstream, making it especially useful for people who may have difficulty sticking to a daily medication routine. However, because it is preventive rather than curative, it does not eliminate HIV in people who are already infected and must be used alongside other protective measures for full sexual health safety.